Abstract
Background Targeting mucosal immunity of the gut, which is known to provide antigen processing, while avoiding excessive or unnecessary inflammation, was tested as a way to modulate COVID-19 severity. <br>
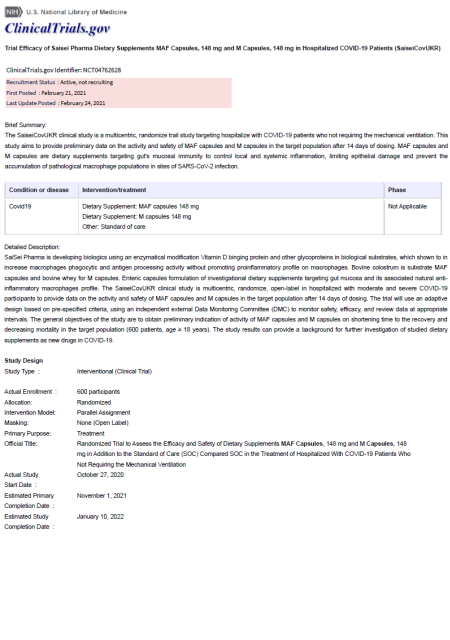
Methods Randomized open-label trial in 204 adults hospitalized with non-critical COVID-19 who received for 14 days in addition to standard of care (SOC) degalactosylated bovine glycoproteins formulations of either MAF capsules (MAF group) or M capsules (M group) or SOC only (control group).

Based on a growing body of evidence that a dysregulated innate immune response mediated by monocytes/macrophages plays a key role in the pathogenesis of COVID-19, a clinical trial was conducted to investigate the therapeutic potential and safety of oral macrophage activating factor (MAF) plus standard of care (SoC) in the treatment of hospitalized patients with COVID-19 pneumonia. Ninety-seven hospitalized patients with confirmed COVID-19 pneumonia were treated with oral MAF and a vitamin D3 supplement, in combination with SoC, in a single-arm, open label, multicentre, phase II clinical trial.

The effects of degalactosylated whey protein on lipopolysaccharide (LPS)-induced inflammatory responses in mice were observed in comparison with intact whey protein. Intraperitoneal administration of both intact and degalactosylated whey proteins for 5 days did not affect body weight and food intake in mice. On day 6, intraperitoneal administration of LPS induced a marked decrease in body weight 4 h later.
The hypothesis: Based on the aforementioned findings and on documented analogies between SARS-CoV-2 and HIV, we hypothesized that the reduced conversion activity of the Gc protein (human group-specific component (Gc)) into the macrophage activating factor (MAF) could have a key role in the dysregulate immune response induced by SARS-CoV-2, just like for HIV infected patients. If this hypothesis is correct, it might help to set a valid strategy of immunotherapy also based on an off-label use of GcMAF in critically ill COVID-19 patients.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7513798/
Conclusion: According to the provided literature overview, we firmly believe that GcMAF deserves be tested as immune-therapeutic to increase macrophages functionality for earlier SARS-CoV-2 viral control, protection against COVID-19 progression by limiting epithelial damage, control local inflammation and prevent from the hyperinflammatory immune response. For this purpose, we planned a Phase-II interventional clinical trial evaluating the effectiveness and safety of Oral immunotherapy with Third Generation GcMAF in hospitalized patients with COVID-19 pneumonia (COral-MAF1 Trial) at the “Ospedale del Mare” Hospital, Naples, Italy.

Development of colostrum MAF and its clinical application -


2016 Case Report: GcMAF Treatment in a Patient with Multiple Sclerosis (PDF)
ANTICANCER RESEARCH 36: 3771-3774 (2016)
学会発表資料の掲載

医療法人再生未来の乾利夫理事長は、2014年12月13日に香川県県民ホールで開催された、第18回バイオ治療法研究会学術集会で、『末期ガンに対するソノダイナミック治療と初乳MAFの併用 』について発表をいたしました。
発表資料ダウンロード
バイオ治療法研究会(公式サイト)
大学との共同研究と研究論文


医療法人再生未来の乾利夫理事長は、2014年7月20日に第5回癌先進補完医療研究会で、コーリーワクチン、2nd GcMAF、オゾン療法の症例等を発表いたしました。
発表資料ダウンロード

医療法人再生未来の乾利夫理事長は、2014年6月27~29日にドイツ ベーヴェルンゲンで開催された第9回医療用レーザー国際会議で、2nd GcMAFの症例等を発表いたしました。
発表資料ダウンロード
医療用レーザー国際会議公式サイト

癌の根治と医学の発展のため下記の大学と共同研究を進めております。